RESEARCH AREAS
Ion Transport Through Nanopores
Electrical Double Layer Capacitors (EDLCs)

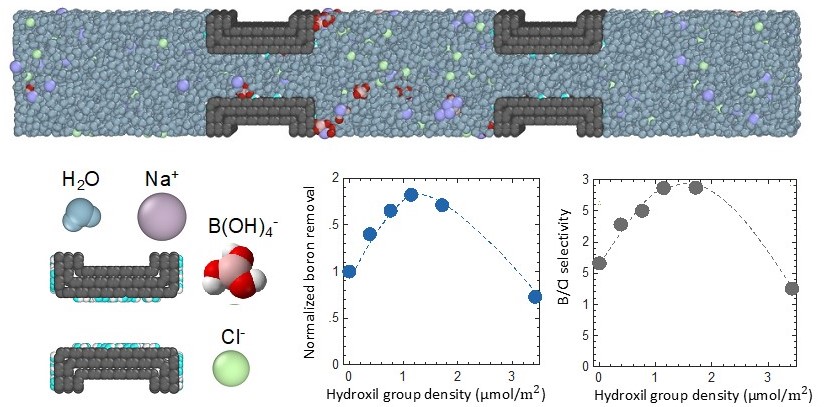
Capacitive Deionization (CDI)
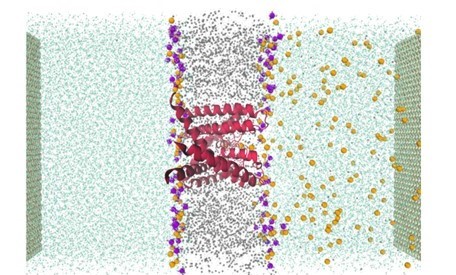
Biological Ion Channels
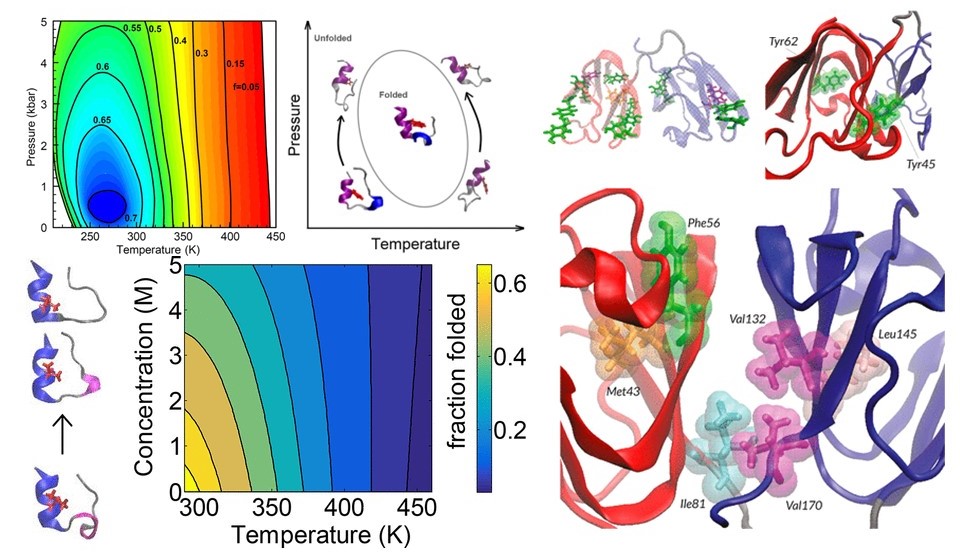
Conformational Transitions in Proteins
Protein Folding Kinetics
Proteins constantly shift between folded, partially folded, and unfolded states. We use
advanced sampling techniques to map the energy landscapes that govern these transitions
and to identify the molecular events that trigger folding or unfolding. Our work illuminates
why some proteins remain stable under extreme conditions while others misfold, offering
clues into the origins of diseases linked to protein instability.
Conformational Transitions Under Environmental Perturbations
Temperature, pressure, and chemical environments can dramatically reshape a protein’s
structure. Through detailed simulations, we quantify how these perturbations alter folding
pathways revealing, for example, how cold, heat, or high pressure can converge to produce
similar unfolded states. These findings help connect fundamental thermodynamics with
practical challenges in biotechnology, biophysics, and drug formulation.
Mutational Dynamics in γD Crystallins
Human γD-crystallin is a long-lived eye-lens protein whose destabilization is associated with
cataract formation. We study how mutations or environmental stresses reorganize its
conformational landscape, revealing hidden intermediate states that may act as precursors
to protein aggregation. This molecular insight strengthens our understanding of age-related
eye diseases and informs efforts to design stabilizing strategies.
Complex Phase Behavior in Fluids
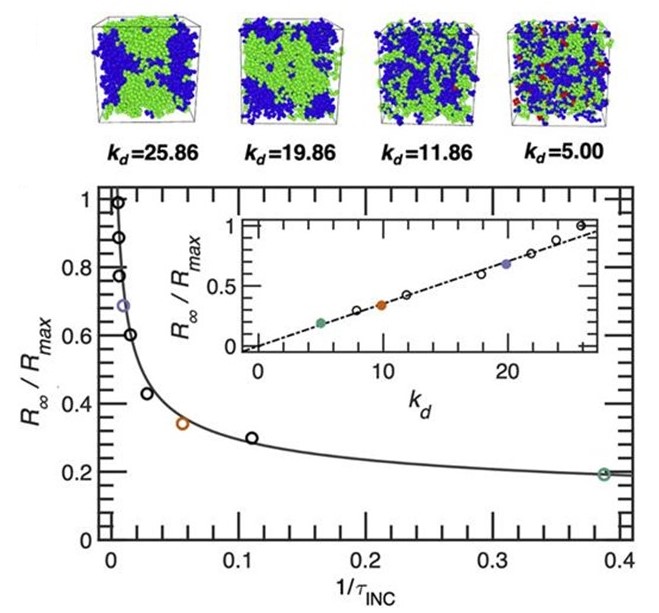
Phase Behavior in Chiral Mixtures